Plant Lab
Testing Plant Substances as Potential Medicines
Background: All plants can be infected by viruses and bacteria, but many of them have a defense mechanism. Isolating this antimicrobial molecule could give a possible medicine. Finding an active ingredient that has antimicrobial activity is a difficult process. Samples must be collected and tested for their ability to kill microbes and prove that it doesn’t cause bad effects in humans. The process for testing the plants are adding an extract-soaked filter paper disks to bacteria cultures on Petri plates. Then they see if there are clear halos in it. That means it inhibited the bacteria’s growth or killed it.
Purpose/Objective: What plant materials, found locally, contain active ingredients that will inhibit the growth of bacteria?
Materials:
Procedure:
Background: All plants can be infected by viruses and bacteria, but many of them have a defense mechanism. Isolating this antimicrobial molecule could give a possible medicine. Finding an active ingredient that has antimicrobial activity is a difficult process. Samples must be collected and tested for their ability to kill microbes and prove that it doesn’t cause bad effects in humans. The process for testing the plants are adding an extract-soaked filter paper disks to bacteria cultures on Petri plates. Then they see if there are clear halos in it. That means it inhibited the bacteria’s growth or killed it.
Purpose/Objective: What plant materials, found locally, contain active ingredients that will inhibit the growth of bacteria?
Materials:
- Balance, weigh boat, lab scoops
- LB broth base
- Media bottles, 250 mL
- Sterilizer/autoclave
- Water bath
- Sterile LB agar
- Laminar flow hood and disinfectant
- Glasses
- Bunsen burner and gas lighter
- Inoculating hoop
- Petri dishes
- E. coli
- Plant specimen
- mortar and pestle
- Pipet
- Plastic funnels
- Filter paper disks
- Beakers
- Syringe
- Reaction tubes and rack
- Methanol
- Dry block heater
- Forceps
- Ampicillin
- Glass spreader
- Incubator
Procedure:
- The profilter was attached to a syringe and “rinsed” with H2O. Next the sterilization filter was opened, with the filter kept in its plastic covering. Approximately 1.5 mL of H20-based filtrate was loaded using a pipet. The plunger was used to collect the sterile filtrate into the microfuge tubes. The microfuge tubes were then quickly closed until a "snap" sound was heard.
Preparing Plant Extracts:
The mortar and pestle was used to grind up 2 grams of plant tissue with 10 mL of deionized water. The deionized water was let sit for 3 minutes and the sample was filtered through an 11 cm filter paper funnel. The filtered sample extract was filter sterilized using a syringe filter. 1 mL of extract was collected into a 1.7 mL microtube. These steps were then repeated, but the water was replaced with methanol as the extracting solvent.
Final Procedure:
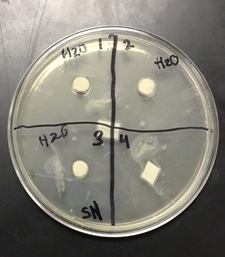
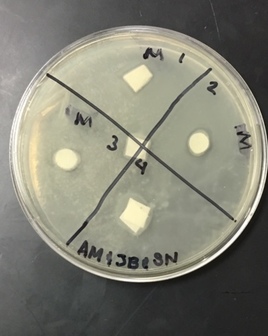
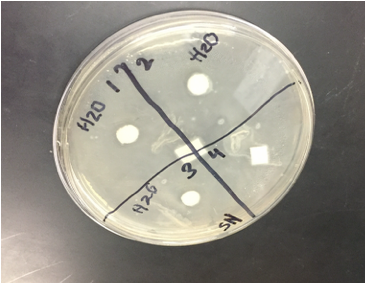
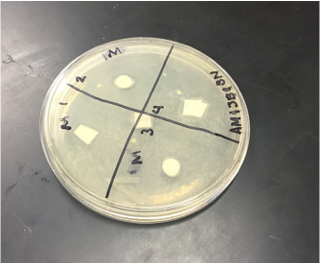
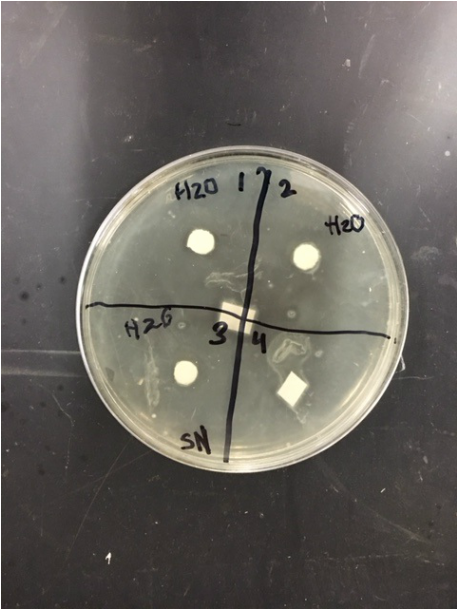
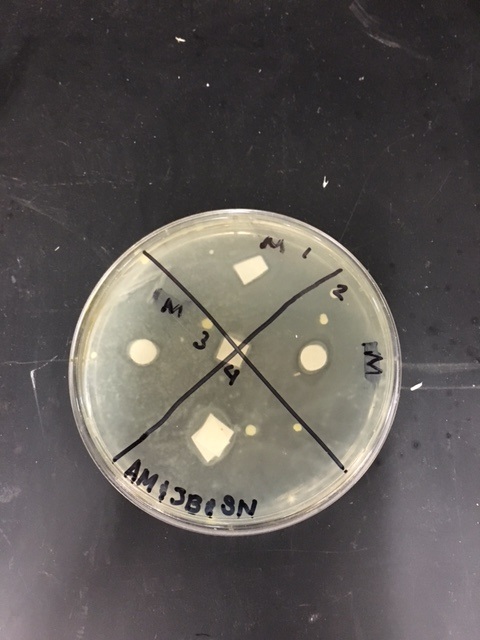
Sterile foreceps were used to add sterile disks to tubes containing 1 mL of sterile water and 1 mL of ampicillin. 10-20 mLs of warmed, pressure sterilized nutrient agar was poured into 2 petri dishes, using sterile technique. After allowing the agar to solidify, plates were turned upside down and sterilized at 4º Celsius overnight. One mL of E.coli colony was added to each plate. A flame-sterilized spreading loop was used to spread the bacteria evenly throughout the surface of the agar. Using sterilized forceps, filter disks were placed in separate quadrants onto the plate in the following sequence: 1) water 2) plant extract 3) ampicillin. Plates were left on the lab bench for 20 minutes to allow bacteria and filter disks to adhere to the agar. Plates were then incubated upside down overnight in a 37º Celsius incubator. Plates were then photographed and observed for clearings around the filter disks after 24 hours, 48 hours and 72 hours. Any bacterial or fungal contamination was noted.
Results: After 24 hours we noticed that there was bacterial growth that contaminated our petri dish with H2O in it. The plate must have been contaminated during our procedure as a result of human error. There was a clearance in the ampicilin area however. After 72 hours, bacteria contaminated in our H2O and methanol dishes. There was contamination in all our dishes which gave us inaccurate results. During our 72 hour observation our group could see continued growth in the bacterial contamination and that there continued to be clearance in every quadrant 4.