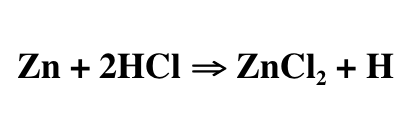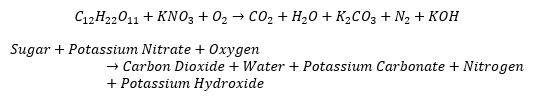Chemical Dominoes
For our project chemical dominoes, we were given a task to design a board game for an of audience of children ages 10+ incorporating various aspects of chemistry in our game. At the least we had to include double replacement, single replacement, a production of gas, chemical and physical (mechanical) changes of energy as well as the lighting of the LED. With those components are also blueprint drawings of the board game, playing instructions, safety precautions and detailed explanation of each chemical reaction.
Content
For our chemical dominos, we created a pirate themed board game. Players would start at a corner of the board. Rolling a dice, they would move that amount of spaces before running into a stop, distinguished as a treasure chest. At each stop we had an activity that the player would have to complete before being able to move. The objective of the game is to reach the middle island first. The first one to reach the island will be able to get the treasure (an LED light). In order to light the LED, we designed a circuit that would only light up the LED if the strip of potassium nitrate and sugar is burned, disconnecting the aluminum foil strip that completes the circuit. By breaking that link, the LED will light up, signifying the victory of a player.
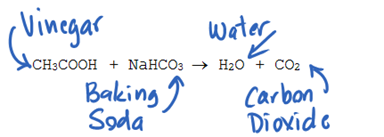
At one of the stops, a player had to make a balloon rise before being allowed to move forward. To fill up the balloon, one would create a gas by strapping a balloon full of bicarbonate (baking soda) on to the top of a graduated cylinder of acetic acid (vinegar). The chemical reaction will create a gas, carbon dioxide and water. The carbon dioxide would rise in the balloon, making it rise.
.
At one of the stops, a player had to make a balloon rise before being allowed to move forward. To fill up the balloon, one would create a gas by strapping a balloon full of bicarbonate (baking soda) on to the top of a graduated cylinder of acetic acid (vinegar). The chemical reaction will create a gas, carbon dioxide and water. The carbon dioxide would rise in the balloon, making it rise.
.
At another stop, the player would have to place a piece of zinc in a cup of hydrochloric acid (a group member wearing appropriate safety gear would do this part). This would be a single replacement reaction where the hydrogen in the hydrochloric acid would be replaced with the zinc, creating zinc chloride and a by product of hydrogen. The hydrogen gas can be seen as the bubbles in the acid. The bubbles are an indicator of chemical change.
Finally we had a stop where the player would test three different salts to see which one would give off a bright red flame. The player would dip a wood stick in water and dip it in one of three salts: strontium, potassium, and. Then one our group members would light the salt on fire. If the salt gave off a bright red flame (which would be strontium), then the player would be allowed to move forward.
To disconnect the circuit and light the LED, we burned a mixture of potassium nitrate and sugar. The chemical reaction releases a cloud of smoke of carbon dioxide, potassium carbonate, nitrogen, and potassium hydroxide as well as water. This shows double replacement reaction, production of a gas, chemical and physical changes of energy.
Double Replacement: When the two compounds react, the positive ions (cation) and the negative ions (anion) of the two reactants switch places which forms a new compound.
AB + CD -> AD + BC
Single Replacement: When an element and a compound react, an element takes a place of another element in the compound.
AB + C -> AC + B
Chemical Change: any change that results in the formation of new chemical substances
Examples: color change, formation of a precipitate, formation of a gas, odor change, temperature change
Physical Change: any change that affects the form of a chemical substance, but not its chemical composition
Examples: folding a piece of aluminum foil, molding clay into a new shape, water evaporating, butter melting
AB + CD -> AD + BC
Single Replacement: When an element and a compound react, an element takes a place of another element in the compound.
AB + C -> AC + B
Chemical Change: any change that results in the formation of new chemical substances
Examples: color change, formation of a precipitate, formation of a gas, odor change, temperature change
Physical Change: any change that affects the form of a chemical substance, but not its chemical composition
Examples: folding a piece of aluminum foil, molding clay into a new shape, water evaporating, butter melting
Reflection
Our project went fairly smoothly. We were efficient and good with time management as well as dividing up the tasks. Half of our group worked on the chemistry part of the board game such as figuring out what reactions we wanted and how we wanted to show it. The other half of the group focused on the building of the board game and creating blueprints, game cards, and overall appearance. I felt like our time management and division of tasks was very efficient. What I wish we could have done was go over the instructions one last time because we were missing a couple of parts: the blue prints and safety precautions on hard copy. Those two were done last minute. We worked fairly well together, but there were some times were I felt like we weren't on the same page. However, by talking it out, we managed to find a middle ground. In this group, there were people who led more, but I feel like I should have taken more initiative. I wish that I could have provided more ideas and taken more initiative, rather than simply following what my other group mates told me to do. Two peaks from are project was definitely the outcome and our time efficiency. Two pits were missing parts and a little bit of hiccups figuring out which reactions to use. Overall, this was an extremely fun project to do.